Key Clinical Trials
Teriparatide: Clinical evidence
Reduction in fracture rate and back pain and increased Quality of Life in postmenopausal women treated with Teriparatide
- Objective: To examine the effectiveness of Teriparatide in postmenopausal women with osteoporosis treated for up to 18 months in normal clinical practice in eight European countries
- Study design: 1,648 postmenopausal women with a diagnosis of osteoporosis who were about to initiate Teriparatide treatment were enrolled.
- Overall risk of incident fractures during treatment with Teriparatide
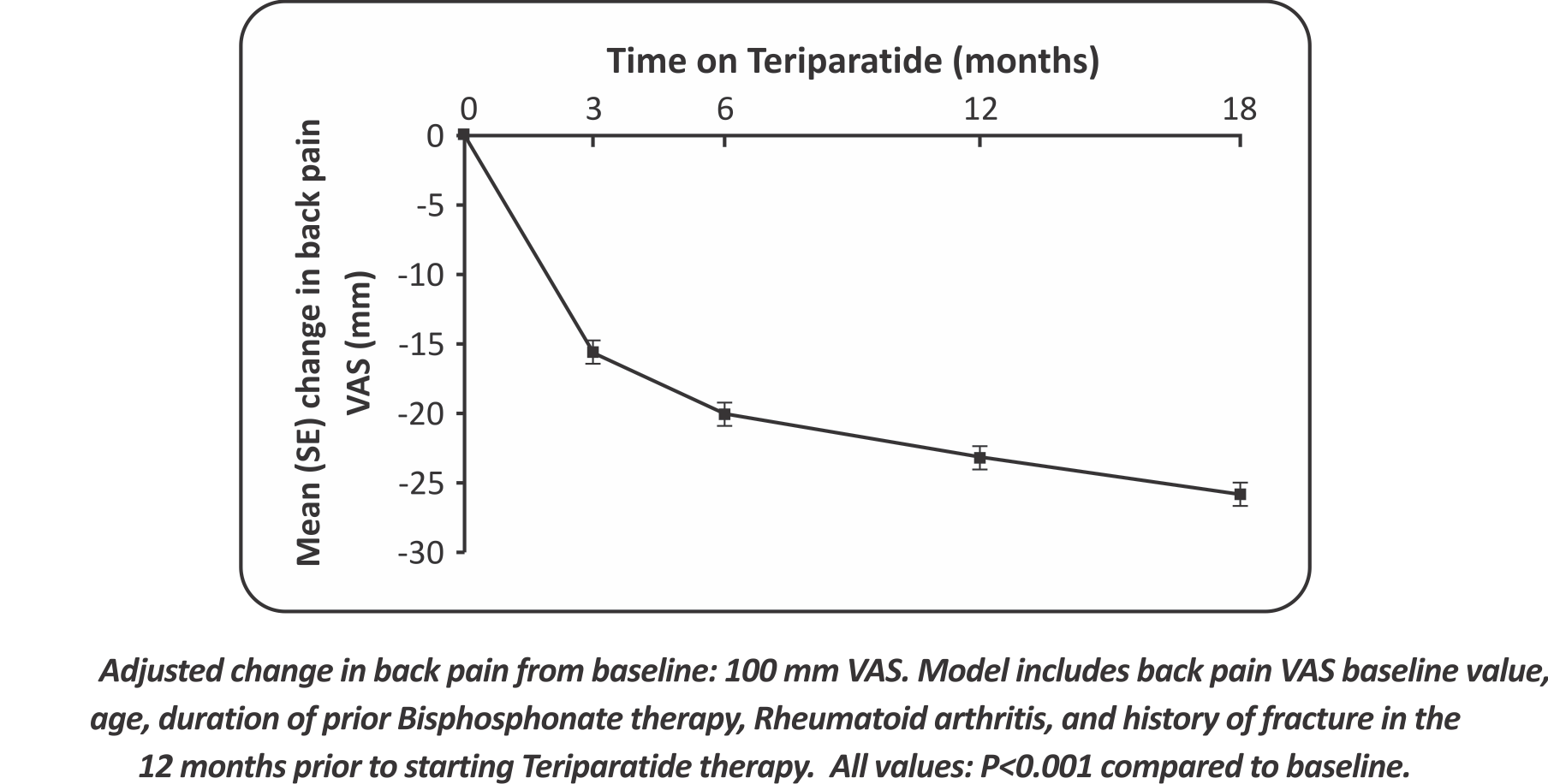
- Adjusted change in back pain from baseline: 100 mm VAS. Model includes back pain VAS baseline value, age, duration of prior Bisphosphonate therapy, Rheumatoid arthritis, and history of fracture in the 12 months prior to starting Teriparatide therapy. All values: P<0.001 compared to baseline.
Changes in Health Related Quality of Life
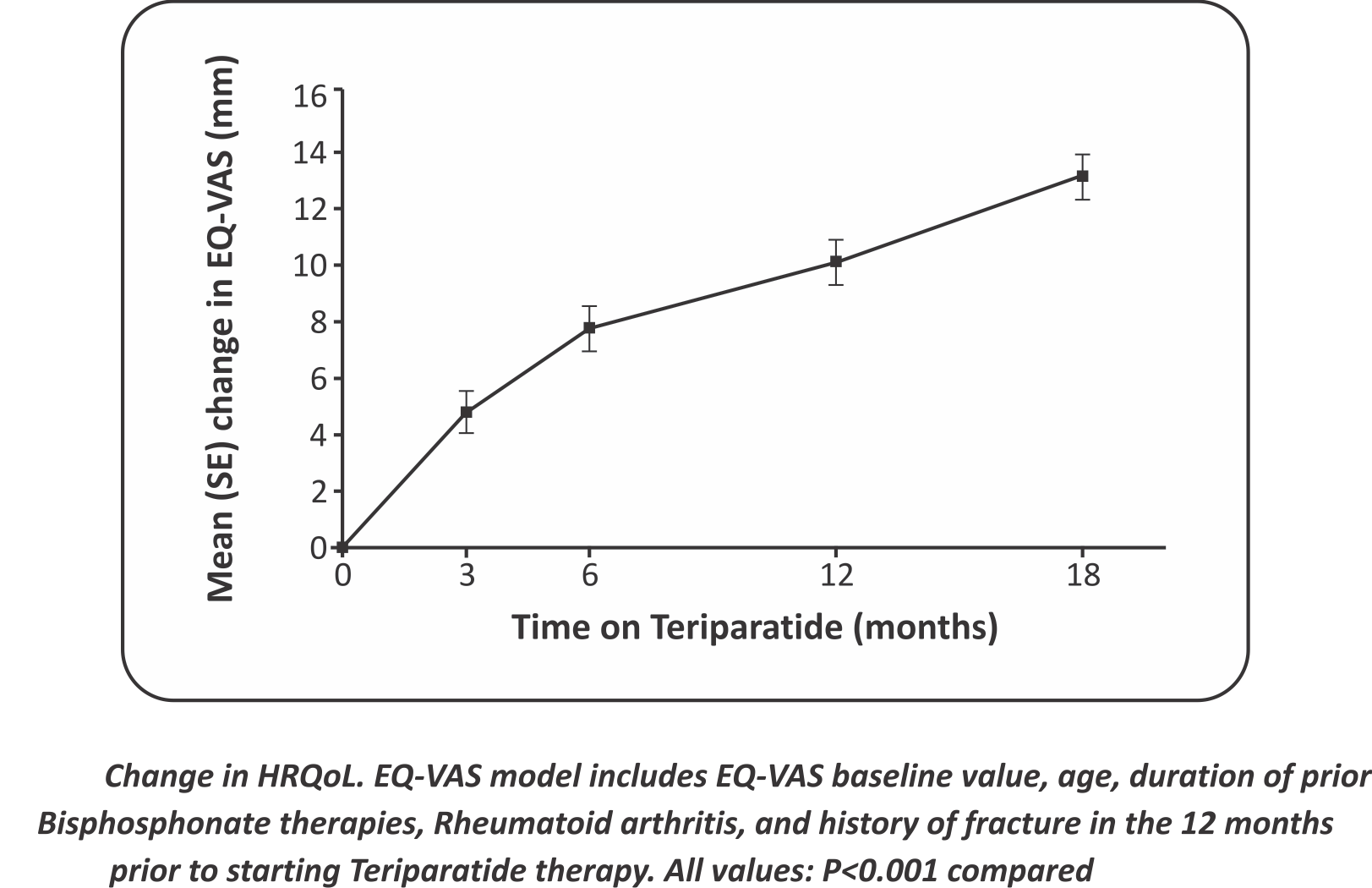
- Change in HRQoL. EQ-VAS model includes EQ-VAS baseline value, age, duration of prior Bisphosphonate therapies, Rheumatoid arthritis, and history offracture in the 12 months prior to starting Teriparatide therapy. All values: P<0.001 compared to baseline
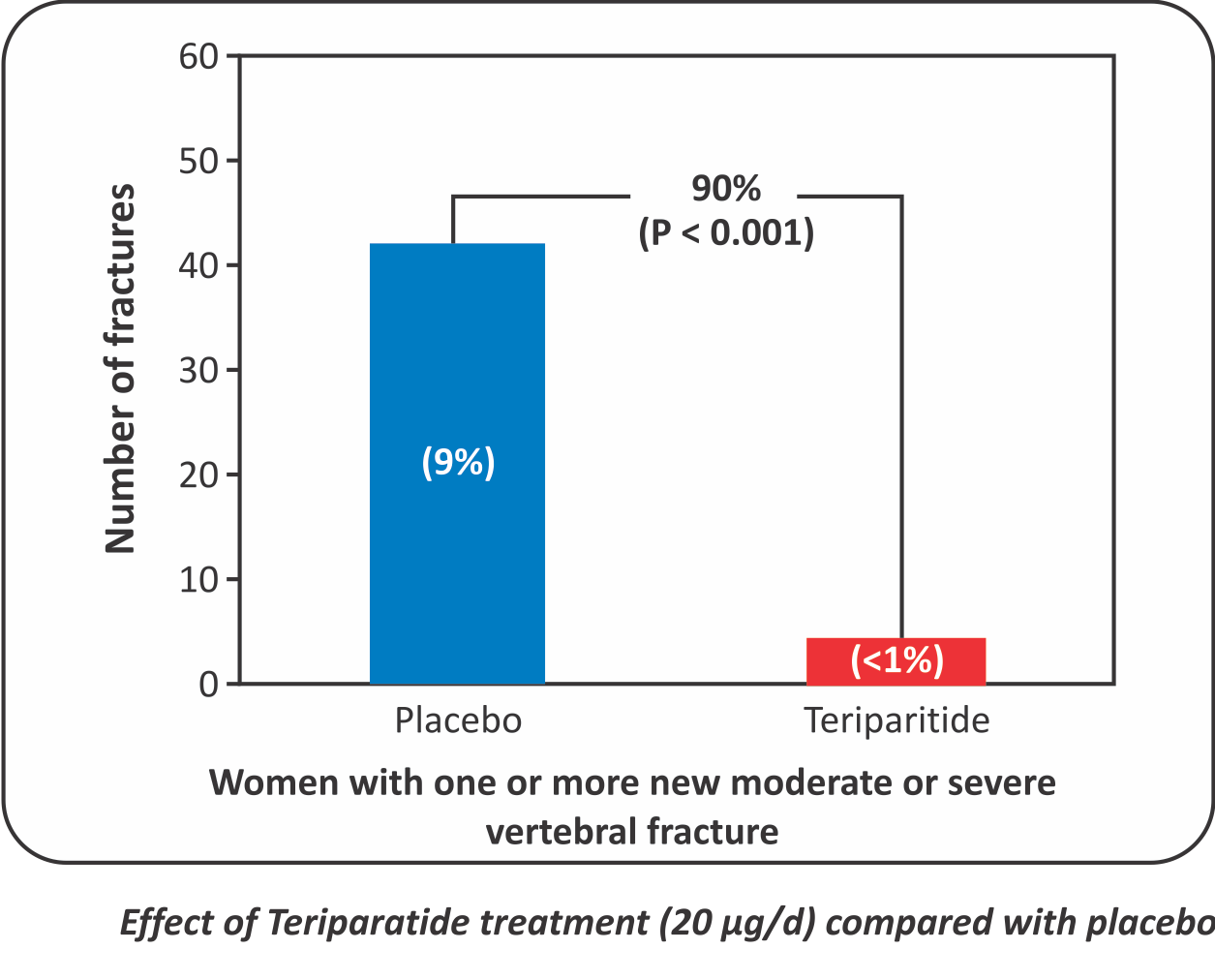
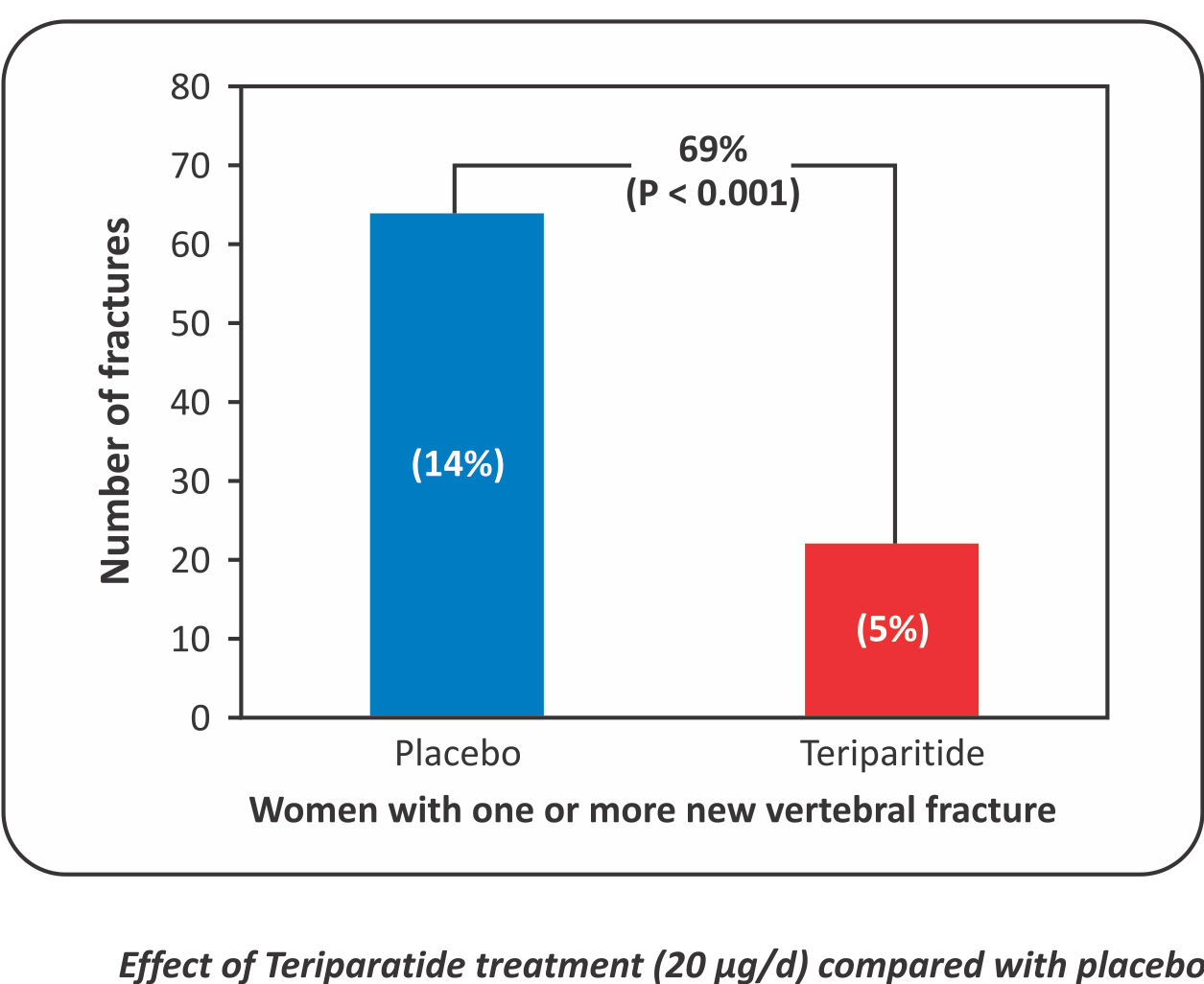
Teriparatide therapy causes 90% reduction in new severe vertebral fractures”
- Background: Once-daily injections of parathyroid hormone or its amino-terminal fragments increase bone formation and bone mass without causing hypercalcemia, but their effects on fractures are unknown.
- Methods: 1637 postmenopausal women with prior vertebral fractures were assigned to receive 20 or 40 pg of parathyroid hormone (1-34) or placebo, administered subcutaneously by the women daily. Vertebral radiographs were obtained at base line and at the end of the study (median duration of observation, 21 months) and performed serial measurements of bone mass by Dual-Energy X-ray Absorptiometry.
Results
Teriparatide: In non-union fracture cases, 93 percent patients show healing and pain control with only 8-12 weeks
- Researchers at the University of Rochester Medical Center in NY, USA gave Teriparatide, to 145 people who had bone fractures that had not healed, many for 6 months or more. They found that
- 93% of patients showed significant healing and pain control after 8-12 weeks.
- They also found that Teriparatide speeds the healing of fractures by changing the behavior and number of cartilage and bone stem cells involved in the healing process, cutting healing time by more than half.
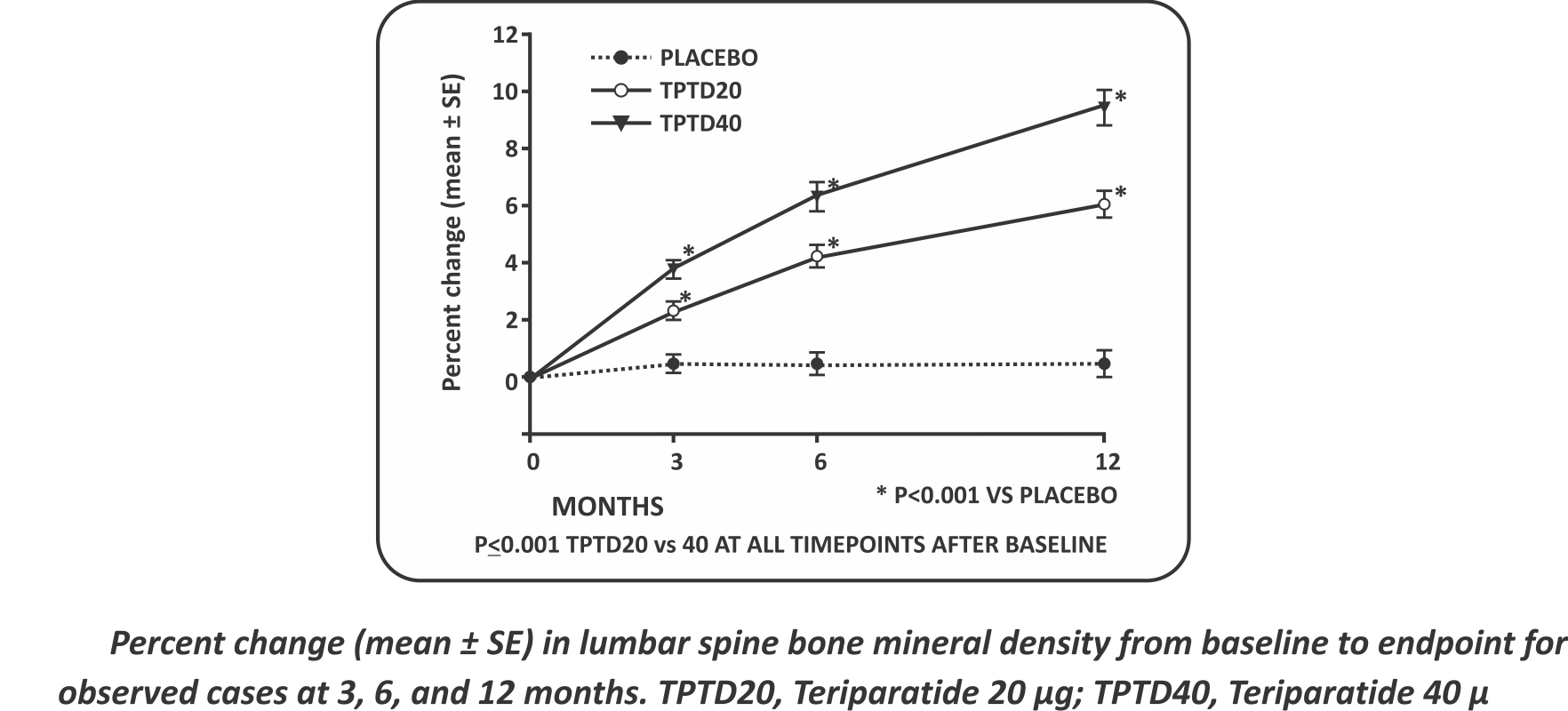
- A linear increase in lumbar spine BMD was observed in the treatment group with Teriparatide in male Osteoporosis
- Introduction: Teriparatide [rhPTH(1—34)] increases bone mineral density and reduces the risk of vertebral fracture in women.
- Method: 437 men were randomized with spine or hip bone mineral density more than 2 SD below the young adult male mean to daily injections of placebo, Teriparatide 20 pg, or Teriparatide 40 pg. All subjects also received supplemental Calcium and Vitamin D. Men were recruited at 37 centers in 11countries from hospital clinics and community practices. Subjects were eligible if they were aged 30—85 years, ambulatory, free of chronic, disabling conditions other than osteoporosis, and had lumbar spine or proximal femur (neck or total hip) BMD at least 2 SD below the average for young, healthy men (lumbar spine: 0.871 g/cmz for Hologic densitometers and 0.980 g/cmz for Lunar densitometers). Investigators were encouraged to enroll equal numbers of subjects with low and normalfree testosterone.
Results
Clinical efficacy of Teriparatide Vs Alendronate
- Methods: In an 18-month randomized, double-blind, controlled trial, we compared Teriparatide with Alendronate in 428 women and men with Osteoporosis (ages, 22 to 89 years) who had received glucocorticoids for at least 3 months (prednisone equivalent, 5 mg daily or more). A total of 214 patients received 20 pg of Teriparatide once daily, and 214 received 10 mg of Alendronate once daily. The primary outcome was the change in bone mineral density at the lumbar spine. Secondary outcomes included changes in Bone Mineral Density at the total hip and in markers of bone turnover, the time to changes in Bone Mineral Density, the incidence of fractures and safety.
Results
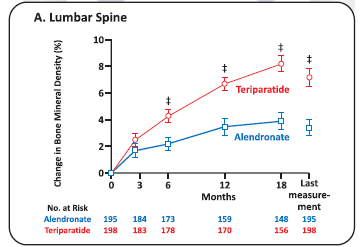
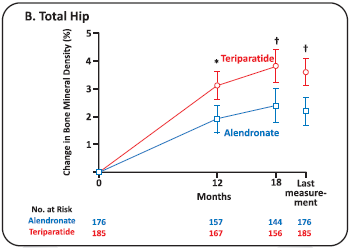
- The *denotes P<0.05, Tdenotes P<0.01, and the idenotes P<0.001 for between-group comparisons. Within-group changes from baseline at the lumbar spine (Panel A) and total hip (Panel B) were significant at all time points (P<0.001). The I bars represent standard errors.
- Conclusions: Among patients with Osteoporosis who were at high risk for fracture, bone mineral density increased more in patients receiving Teriparatide than in those receiving Alendronate.
Teriparatide: Clinically proven better in postmenopausal women with Osteoporosis
- Background and objective: Osteoporosis is emerging as a leading cause of substantial morbidity in India, particularly in postmenopausal women. Teriparatide (rhPTH [1-34]) increases bone formation and improves bone micro-architecture, thereby reducing the risk of fractures. This study was conducted to evaluate the efficacy of Teriparatide in increasing Bone Mineral Density (BMD) in postmenopausal women with Osteoporosis.
- Material and methods: A randomised, prospective, multicentre, open-label, controlled study was conducted on 82 postmenopausal women with established osteoporosis. Patients were randomly divided into control and Teriparatide groups, each group consisting of41 patients. All the patients were supplemented with 1000 mg of elemental calcium and 500 IU of vitamin D throughout the study period of 180 days. Besides, Teriparatide group patients were administered Teriparatide 20pg daily subcutaneously. Lumbar spine, femoral neck and total hip BMD, Bone Mineral Content (BMC) and bone area were measured by Dual Energy X-ray Absorptiometry (DXA) at baseline and at the end of 6 months of treatment.
Results
- The investigational product Teriparatide was well tolerated and there were no serious adverse events. In addition, there were no significant differences between the groups in the incidence of adverse events.
- The percentage of increase in lumbar spine BM D, which is the primary endpoint, was significantly (P < 0.001) higher in Teriparatide group compared to that in control group (6.58% vs. 1.06%)
- Teriparatide significantly increased percentage of change in lumbar spine T-score (P < 0.001), BMC(P < 0.001) and bone area (P < 0.028) compared to control group at 6 months
- Conclusions: These results show that Teriparatide is an effective and safe drug in increasing the BMD and therefore, Teriparatide provides yet another new therapeutic option for reducing the risk management of Osteoporosis in postmenopausal women.
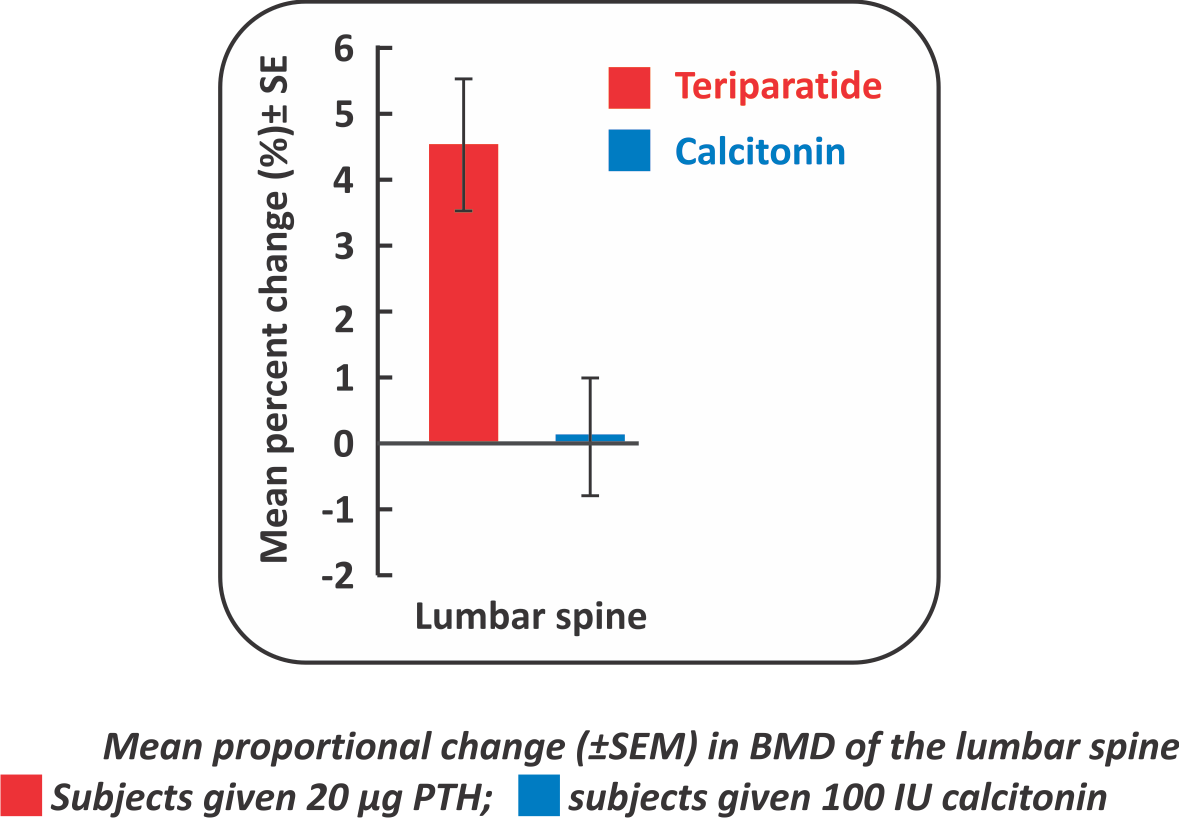
Teriparatide has more positive effects on bone formation than Salmon Calcitonin, as shown by the larger increments of lumbar spine BMD and bone formation markers”
- Introduction: This study compared the clinical efficacy, safety, and tolerability of daily subcutaneous injections of Teriparatide and Salmon Calcitonin in the treatment of postmenopausal women with established Osteoporosis in Taiwan.
- Material and methods: The study included a 6-month treatment phase after a screening period of up to2 months; the patients were randomized to receive either Teriparatide (n=34) or Calcitonin (n=29).Spine and hip BMD were measured at baseline and at the end of the study. Biochemical markers of bone metabolism were assessed at baseline, 3 months and 6 months, at approximately the same time in the morning. The centers recruited postmenopausal (at least 3 year after menopause) women with established osteoporosis. Patients were required to have a BMD value as measured by Dual Energy x -ray Absorptiometry, 2 standard deviations below the mean of young women or lower, based on the manufacturer's reference values (spine <0.827 g/cmz, femoral neck <O.695 g/cmz) and a radiographically documented, prevalent osteoporotic vertebral or nonvertebral fragility fracture.
Results
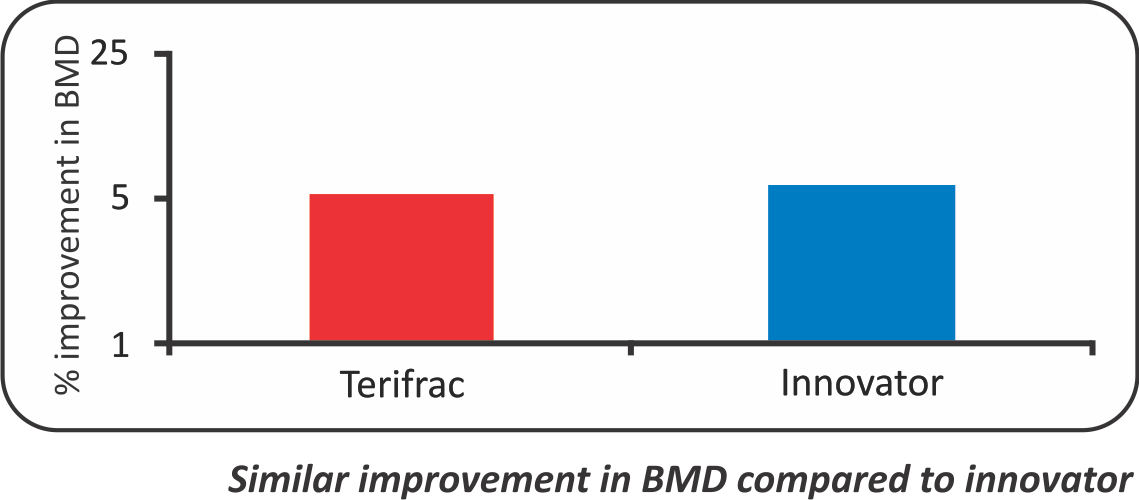
- Safety and efficacy of Terifrac (lntas Teriparatide) [rhPTH (1-34)] against innovator (Forteo of Eli-Lilly) in postmenopausal females with Osteoporosis - A biosimilarity study“
- Objectives: The purpose of the study is to prove Teriparatide [rHu-PTH] of Intas Biopharmaceuticals Ltd is as efficacious and safe as lnnovator's product.
- Primary objective of this study is to compare the efficacy of Teriparatide [rHu- PTH(1-34)] of Intas Biopharmaceuticals Ltd. against Forteo of Eli-Lilly in postmenopausal females with Osteoporosis
- Secondary Objective ofthe study is to collect and compare the safety data ofTeriparatide [rHu-PTH] of Intas Biopharmaceuticals Ltd. with that of Forteo of Eli Lilly.
Efficacy Conclusion: (p value<0.05)
- After Treatment for 3 Months, the mean increase in BMD (L1-L4) values was 0.77 to 0.81 in Intas Teriparatide group and from 0.78 to 0.83 in forteo. (p value<0.05)
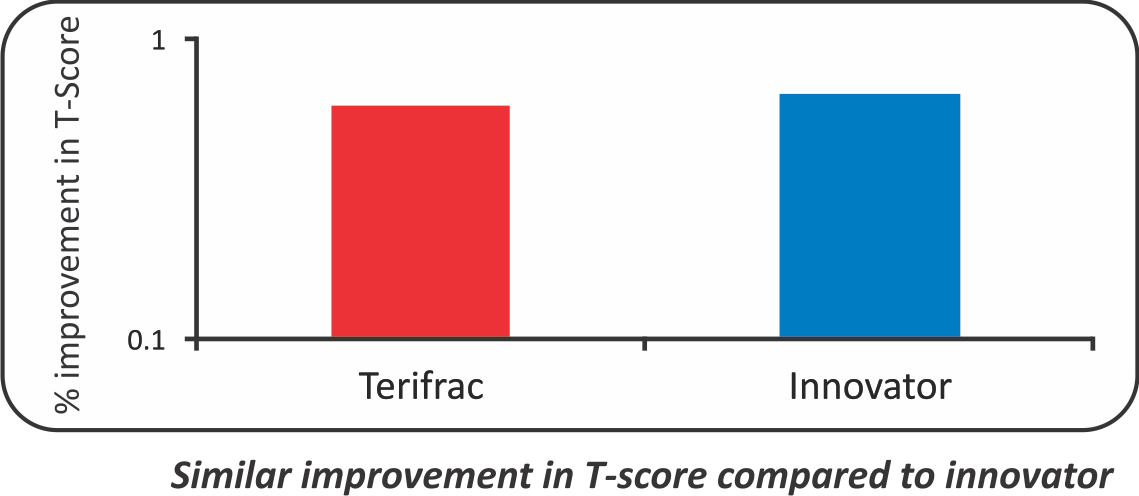
- The mean increase in T Score (L1-L4) was -3.41 to -3.11 in Intas Teriparatide group and -3.28 to -2.91 in Forteo Group. (p value<0.05)
- The mean BSAP increased from 20.45 to 27.92 Intas Teriparatide group and from 18.63 to 26.75 in Forteo group i.e. 47.18% and 48.55% change respectively. (p value<0.05)
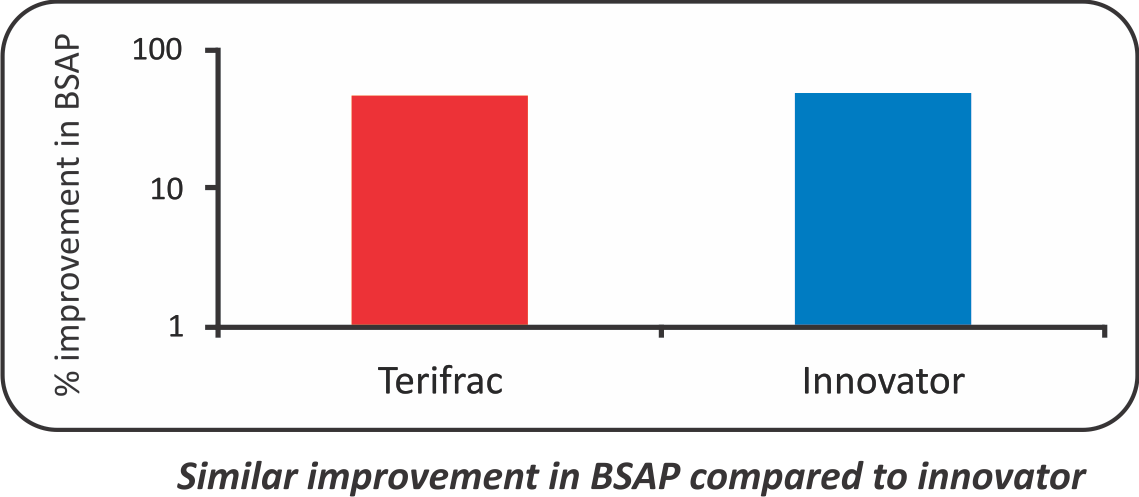
- BSAP - Bones Specific Alkaline Phosphatase
Safety Conclusion
- There was no any adverse event reported due to abnormal laboratory values during study.
- All laboratory parameters were within acceptable range during the study.

